CAUSTIC SODA
WHAT IS CAUSTIC SODA?
Caustic Soda is the name given to the chemical makeup of sodium hydroxide. This material is an alkali, a kind of base that can neutralize acids, is soluble in water, and has the attribute of being basic. Currently, caustic soda can be made in a variety of forms, including pellets (granules), flakes, powders, liquids, and more. The corrosive base, or caustic soda, which is a sort of chemical component, is used by a broad range of various types of enterprises. Every nation is keen to import this chemical since it is a necessary component of the manufacturing of a broad range of products, including textiles, paper, soaps, and cleaning products.
Content Overview
We export the highest-quality Caustic Soda / Sodium Hydroxide in different types and grades from Iran, Turkey, and the UAE to other countries around the world. (Petronaft is a shareholder of two large caustic soda manufacturer companies.)
- Iran Caustic Soda / Iran Sodium Hydroxide
- Turkey Caustic Soda / Turkey Sodium Hydroxide
- UAE Caustic Soda / UAE Sodium Hydroxide
PHYSICAL PROPERTIES OF CAUSTIC SODA
• It has the capacity to absorb a substantial quantity of carbon dioxide and water from the environment around it.
• In its purest form, it takes on the appearance of a crystalline solid that is white in color. This is the condition in which it may be discovered.
• It has a viscosity of 78 megapascal, which is more than that of water; hence, it is a material that is more viscous than water.
• Having a density of 2.13 grams per milliliter and a melting point of 318 degrees Celsius, it may be melted at these temperatures. In addition to that, there is no discernible smell about it.
• The three solvents that have the least amount of trouble dissolving it are water, ethanol, and glycerol in that order.
• When it is dissolved in water, it triggers a reaction that is very high in exothermic activity.
• It is able to produce hydrates in sufficient quantities.
• It is available to purchase in both solid and liquid form.
CAUSTIC SODA PRODUCTION
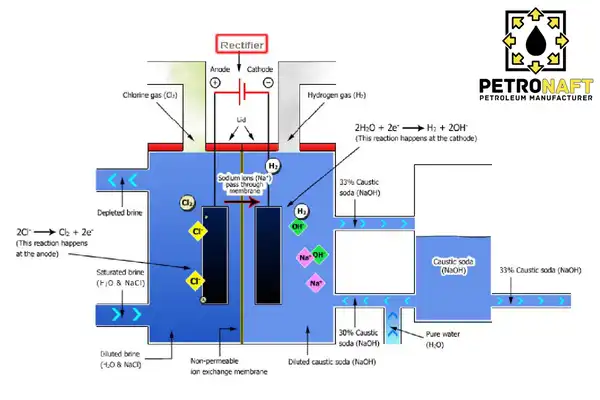
Caustic soda, also known as sodium hydroxide, is normally produced by electrolyzing a solution of sodium chloride, more often known as brine. This specific approach is referred to by its more common term, the chloralkali process. The electrolysis of brine is an essential stage in the production of chloralkali. This step takes place in a cell that consists of two independent chambers that are partitioned off from one another by a porous diaphragm. As a result of the flow of electricity through the cell, the sodium ions in the brine are attracted to the cathode, whilst the chloride ions are pulled to the anode. One of the chambers of the cell is where the brine is introduced.
At the cathode, chloride ions are oxidized, which results in the production of chlorine gas. At the anode, sodium ions are reduced, which results in the formation of sodium metal. The sodium metal and chlorine gas that are produced during this process are then mixed with water in order to produce hydrochloric acid and caustic soda. It is essential to keep in mind that the chloralkali process requires a significant quantity of energy and carries the risk of producing potentially hazardous byproducts, such as chlorine gas and sodium metal. Both of these components must be managed with the utmost care and attention at all times.
CAUSTIC SODA IN CLEANING PRODUCTS
In many different cleaning solutions, including those intended to clean ovens and drains, caustic soda is a component. Caustic soda is a useful ingredient in these goods since it is corrosive. Because it produces heat by reacting with the moisture in the air, it can change the state of fats and grease to the point where they are dissolved. Despite being FDA-approved and completely safe to use in cleaning solutions, caustic soda shouldn’t be ingested, touched, inhaled, or used in locations with flammable materials.
CAUSTIC SODA IN MANUFACTURING
Caustic soda is used in a variety of sectors, mostly as a powerful chemical base, in the manufacture of drinking water, pulp and paper, soaps and detergents, and drain cleaners. In addition to de-inking and water treatment at pulp mills, the pulp and paper industry also uses caustic soda for pulping and bleaching. The manufacture of rayon and cellophane, soap, and textiles are only a few of the many applications for Caustic Soda, which also includes the extraction of alumina.
CAUSTIC SODA IN WATER TREATMENT
By absorbing both water and carbon dioxide, caustic soda (sodium hydroxide, or NaOH) elevates the pH of water.
The Hard Water:
Corrosion results from “hard” water. Magnesium and calcium dissolved in hard water are in large quantities. In a water treatment plant, minerals cling to the tanks and lines, obstructing water flow and, if untreated, eventually corroding through the line. The saponification of soaps and detergents is known to be reduced by hard water; as a result, clothing get scratchy and discolored, dishes and glasses become discolored, and buildup appears on the skin and appliances. When added to a water treatment system, caustic soda raises the pH of the water and may readily transform hard water into something that is much closer to neutral.
Reduction of Hazardous Metals:
Dangerous metals like lead and copper lose some of their solubility in caustic soda. These metals are heavy and bulky while in their insoluble condition, which increases the possibility that they may get stuck in a filter or fall to the bottom of a tank where they can be collected.
Inhibitor of Corrosion:
In contrast to other additives, which release calcium carbonate as a consequence of sequestration, caustic soda acts as a corrosion inhibitor without causing an uneven distribution of calcium carbonate. Calcium carbonate, despite its capacity to adhere to pipe walls and give some degree of protection against caustic chemicals, inhibits the flow of water due to its excess and uneven distribution, which makes it difficult for water to reach its destination.
It has been shown that the use of caustic soda in the treatment of water needs far less maintenance than the use of other pH adjustors. Caustic soda helps to clean lines and increase water flow by sequestering and saponifying lipids and oils.
1-CAUSTIC SODA FLAKES:
The chemical breakdown of sodium chloride produces Caustic Soda Flakes, a white, water-soluble substance. Caustic Soda Flakes are used in the manufacturing of soaps, shampoos, and fatty substances due to their high hydrolyzing capacity. It is known scientifically as sodium hydroxide. The chemical composition of Caustic Soda Flakes has a high alkaline characteristic and is regarded a PH regulator; it may be used to neutralize acids and make various salts.
Caustic Soda Flakes are a concentrated form of sodium hydroxide in liquid form. Caustic Soda Flakes are a kind of ionic salt composed of sodium ions Na+ and hydroxide ions OH- in the form of NaOH. The solution of Caustic Soda Flakes degrades proteins and, as a consequence, produces burns when it comes in touch with bodily tissues. This substance collects moisture and carbon dioxide gas from the surrounding environment and has a high solubility in water. The yearly output of Caustic Soda exceeds tens of millions of tons due to its enormous demand worldwide.
Normal temperature Caustic Soda is a white solid, but when it comes into contact with acids and is diluted in water, an exothermic reaction occurs and a great deal of heat is created. Caustic Soda is frequently used in the soap manufacturing and petrochemical sectors. It is also employed as a metal detector, in chemical processes, to clean cotton, to remove rust from metal, etc.
CAUSTIC SODA FLAKES SPECIFICATIONS
Pure Caustic Soda Flakes are a solid material that resembles colorless crystals quite closely. It melts without decomposition at 318 °C (604 °F) and boils at 388.1 °C. Although this chemical is highly soluble in water, its solubility in polar solvents is much lower (such as ethanol and methanol). (It is important to note that they are insoluble in ether and other non-polar solvents.)
Similar to the hydration of sulfuric acid, the dissolving of solid Caustic Soda Flakes in water is a highly exothermic process that generates a great deal of heat and poses significant safety risks when handling the substance. As with other alkaline solutions, the saponification that happens between the Caustic Soda Flakes and the skin’s natural oils makes the resultant solution slippery if it comes into contact with the skin. Its viscosity is inversely proportional to its service temperature, meaning that the rise in viscosity is proportional to the increase in temperature. Applications and storage of Caustic Soda Flakes are directly influenced by their viscosity.
CAUSTIC SODA FLAKES APPLICATIONS
- Production of chemicals: Approximately forty percent of Caustic Soda Flakes are utilized as a raw material and foundation for the production of several chemicals in the chemical industry.
- Detergent and disinfection goods Caustic Soda Flakes are used to manufacture soap and different detergents with a variety of domestic and industrial uses. The combination of chlorine with Caustic Soda solution produces chlorine bleaching solutions (chlorine-containing bleaches such as sodium hypochlorite or chlorinated water). Pipe unclogging treatments containing Caustic Soda prevent pipes from clogging by transforming potentially clogging fats and oils into water-soluble soapy compounds.
- Applications of Caustic Soda Flakes in medicine: Caustic Soda Flakes is employed in the synthesis of several pharmaceutical compounds, ranging from basic pain relievers like aspirin to anticoagulants that prevent blood clots and anti-arteriosclerosis medications.
- Pulp and paper production: Pulp generated by the sulfate and sulfite technique is refined by eliminating lignin components using several extraction units of Caustic Soda Liquid. In addition, Caustic Soda Liquid is used in certain paper mills that employ the kraft process. In addition, this chemical compound is utilized in the paper industry to remove ink from recycled paper.
- Cellophane and synthetic silk: The viscose method for producing fibers involves Caustic Soda Flakes in two basic phases. Alkaline cellulose is produced by treating cellulose with Caustic Soda Liquid in order to improve its strength and sheen. The resultant cellulose is then dissolved in diluted Caustic Soda Liquid to generate viscose, which is used to extrude rayon fibers and cellophane films.
- Applications of Caustic Soda Flakes in the manufacture of aluminum: Aluminum extraction is used to dissolve the raw material for aluminum manufacturing, bauxite ore, and precipitate alumina. Also used for chemically polishing aluminum goods is Caustic Soda.
- The fabric: In this business, Caustic Soda Flakes are used for cleaning, bleaching, and improving the fabric’s sheen and tensile strength.
- Industry of oil production and refining: Caustic Soda is utilized as a carbon dioxide absorber in light cuts and as a sulfide absorber in the purification of oil cuts in the oil production and refining business. In addition, it is employed with chlorine in the hypochlorite sweetening process, which removes different sulfur compounds.
- Use in place of Soda ash Caustic Soda Flakes are used in place of Soda ash in a variety of applications in the glass, paper, pulp, phosphate, and silicate industries.
- Applications of Caustic Soda Flakes in the food industry: Food manufacturing; sodium hydroxide is used in the manufacture and processing of a variety of food products. To generate crispness in olives or pretzels, Caustic Soda is used in processing olives or making pretzels. For the purpose of canning, Caustic Soda Flakes are used to peel potatoes, tomatoes, and other fruits. In addition, certain foods are treated with Caustic Soda to inhibit the formation of germs and mold.
- Water and sewage treatment: Caustic Soda Flakes are used in water and sewage treatment facilities to regulate water acidity and remove heavy metals. Also, Caustic Soda is used in the production of the antiseptic sodium hypochlorite.
- Energy: Caustic Soda is employed in fuel cell manufacture. Epoxy resins used in wind turbines are created from Caustic Soda Flakes.
- Renewable fuels: Caustic Soda Flakes are used to regulate pH and create sodium methylate in the bioethanol and biodiesel manufacturing process.
CAUSTIC SODA FLAKES PRODUCTION
Using the hydrolysis of a salt water solution, Caustic Soda Liquid is created in petrochemicals and liquid Soda production units, and while being delivered by special tankers to Caustic Soda Flakes manufacturing units, in special tanks erected inside the unit. The merchandise is discharged. As manufacturing byproducts, units that manufacture Caustic Soda Liquid also generate hydrochloric acid, brine, per chlorine, chlorine gas, and other chemical compounds.
In factories, Caustic Soda Liquid is moved from the main tank to a small air tank before overflowing onto nickel trays and being concentrated at 1400 degrees Celsius from 50% liquid Soda to 98% to 99%. After enough heating, the liquid profit is sent to the cooling drive device and, upon cooling, transforms into thin flakes on the annular wall of the graph. During this procedure, the Caustic Soda Liquid’s water is evaporated, resulting in Caustic Soda Flakes with an analysis of 98% to 99%. Immediately after production, Caustic Soda Flakes are placed in 25-kilogram double-layer bags. This avoids the generated product from absorbing moisture and becoming lumpy.
CAUSTIC SODA FLAKES PACKAGING
Caustic Soda Flakes are offered in a variety of packaging options, including double-walled (laminated) bags with extremely acceptable stitching, as well as palletized and ready-to-ship per client request. The packaging of Caustic Soda Flakes is one of the most significant aspects of manufacturing, shipping, and storage. If the Caustic Soda Flakes are packaged in low-quality bags, air and moisture will enter the bag, causing the flakes to clump and stay together. This diminishes the quality of Caustic Soda Flakes to the point where they are unusable.
The packaging for Caustic Soda Flakes:
- Caustic Soda Flakes are packaged in 25-kilogram bags with dimensions of 50 x 80 and a weight of two layers of 60 × 80 grams each. If ordinary thread is used, Caustic Soda Flakes will rot and disintegrate the sewing thread. Therefore, these bags must be sewed using durable polyester thread. As air enters the bags, Caustic Soda Flakes become lumpy, and the combination of air and sodium hydroxide causes the lumpy substance to expand and finally cause the bag to split and disintegrate. Wherever this sort of Caustic Soda is used, safety and health precautions must be maintained to avoid bodily harm. Pay close attention to the safety guidelines for dealing with Caustic Soda Flakes while handling this substance.
- Jumbo bag: In this packing, Caustic Soda Flakes are packaged in 40 25 kg bags inside a one-ton bag (jumbo bag). The giant bag is a fantastic protection for preventing probable ripping of the bags due to displacement, and it also prevents direct sunlight and air from penetrating damaged bags.
2-CAUSTIC SODA LIQUID:
Sodium hydroxide, sometimes known as Caustic Soda Liquid, is one of the most commonly used bases and a sodium salt. This substance has the chemical formula NaOH and is also known as Caustic Soda Liquid, sodium hydroxide, alkali, alkaline Soda, and sodium hydrate. Caustic Soda Liquid absorbs iron and moisture from the atmosphere, as well as carbon dioxide. The solubility of Caustic Soda Liquid in water, ethanol, glycerol, and methanol is comparable. It should also be mentioned that, due to the rapid absorption of carbon dioxide from the air by this substance, there is a chance of explosive chemical changes occurring in the material during its interaction with carbon dioxide. This normally stable material becomes unstable when heated. When Caustic Soda Liquid comes into touch with the skin, it will have a corrosive impact; thus, the Material Safety Data Sheet (MSDS) must be followed to avoid burns. From solutions of proper composition, sodium hydroxide may be crystallized. Thus, NaOH solutions may be readily cooled to a wide range of temperatures, allowing the formation of hydrates from solutions of varying concentrations. At temperatures over 12.58 °C, however, it often decomposes into solid monohydrate and liquid solution. Unlike most transition metal hydroxides, which are insoluble, sodium hydroxide may be used to precipitate metal hydroxides since it is soluble. At room temperature, a concentrated aqueous solution of 50% sodium hydroxide has an inherent viscosity of 78 mA, which is much more than water (1.0 mA) and olive oil (85 mA). As with other liquid chemical substances, the viscosity of aqueous NaOH is inversely proportional to its service temperature; as the temperature rises, its viscosity reduces, and vice versa. Use and storage of sodium hydroxide solutions are directly affected by their viscosity.
Sodium hydroxide, also known as Caustic Soda Liquid, is a corrosive material that belongs to the family of alkaline substances; hence, specific pipelines and fittings are required for its storage and usage in the manufacturing cycle. Typically, industries use nickel sheet, which has anti-corrosion qualities. To facilitate transport, storage, and application, Caustic Soda Liquid is transformed into Caustic Soda Flakes. Utilizing high-quality Caustic Soda Liquid and modern equipment for the manufacturing of Caustic Soda Flakes from Caustic Soda Liquid results in a product of superior quality and purity.
CAUSTIC SODA LIQUID APPLICATIONS
Caustic soda liquid is used in the manufacturing of paper pulp, aluminum refining, soap, the food sector, water purification, cement, and concrete. Various sectors utilize it as a detergent and industrial grease. Its industrial uses include washing grease, fats, and proteins from tanks and silos, as well as cleaning pipelines and industrial effluent from sediments. It is used to alter the acidity of wells used to extract oil and gas. For basic hydrolysis of esters (such as in soap production), amides, and alkyl halides, sodium hydroxide may be utilized. However, since sodium hydroxide is poorly soluble in organic solvents, potassium hydroxide solution is preferable. Although skin contact with the sodium hydroxide solution is not advised, it generates a slippery sensation. This is caused by the presence of skin oils, such as sebum, which transform into soap. Despite being soluble in propylene glycol, it is unlikely to replace water in soap production owing to the early interaction of propylene glycol with fat, which occurs before the reaction of sodium hydroxide with fat.
CAUSTIC SODA LIQUID PRODUCTION
Using the chlor-alkali method, 50% Caustic Soda Liquid is produced industrially by electrolyzing sodium chloride salt. In the past, this product was produced by the Causticization reaction of sodium carbonate and hydrated lime with the chemical formula calcium hydroxide. This method was supplanted in the 19th century by the Solvay method, and currently the Chlor-alkali method is utilized. Combining pure sodium metal with water is the scientific method for making Caustic Soda. The United States and China are the world’s leading manufacturers of Caustic Soda Liquid. The two industrial procedures utilized by makers of Caustic Soda Liquid to produce sodium hydroxide are:
Diaphragm cell method
Membrane cell method
In the membrane method, which is a novel method for manufacturing Caustic Soda Liquid, a cathode component and an anode component are separated by a membrane inside a single cell. Only sodium ions and a little quantity of water may get through this membrane to the cathode portion. Hydrogen gas is produced and collected as bubbles by the breakdown of water in the cathode portion. The solution of Caustic Soda remains as 30% Caustic Soda Liquid, which may be raised to 50% in subsequent phases. On the other side of the membrane cell, or in the anode, chlorine gas is generated by the breakdown of salt. The membrane cell method for manufacturing 50% Caustic Soda Liquid utilizes the least amount of energy among all industrial processes.
Using a permeable diaphragm, the anode portion is isolated from the cathode portion in the diaphragm method. Concentrated salt water flows from the anode part via the diaphragm to the cathode section. Consequently, the diluted caustic brine is expelled from the cell. In the subsequent procedures, surplus salts may be eliminated to obtain 50 percent Caustic Soda Liquid. In the diaphragm cell method, roughly three tons of steam per ton of Caustic Soda is typically utilized for evaporation. Salt is extracted from caustic brine to raise the product’s concentration. Finally, using specialized techniques, the liquids within may be dehydrated to facilitate storage, packaging, and transport. In the form of Soda Flakes, dry Caustic Soda may be created.
CAUSTIC SODA LIQUID STORAGE
Caustic Soda solution or Caustic Soda Liquid is stable; however, its shelf life is dependent on storage conditions. If this chemical comes into contact with air, the product’s quality will degrade over time, as Caustic Soda absorbs liquid carbon dioxide from the surrounding air and forms solid sodium carbonate (Na2CO3) particles. Therefore, contact between Caustic Soda Liquid and air should be kept to a minimum.
CAUSTIC SODA LIQUID PACKING
Almost we provide Caustic Soda Liquid according to customer inquiries. It is capable to pack in IBC Tanks and drums. IBC capacity will be 1000-1200 kg. Drum capacity is 200-220 kg of Caustic Soda lye.
Explore related articles on this product via the tag link below:
Articles Related to Caustic Soda
Petro naft provides Caustic Soda / Sodium Hydroxide of the best quality in a variety of types and grades and exports them from Iran, Turkey, and the United Arab Emirates to other nations worldwide.
- Caustic Soda Iran / Sodium Hydroxide Iran
- Caustic Soda Turkey / Sodium Hydroxide Turkey
- Caustic Soda Emirates / Sodium Hydroxide Emirates
Understanding Iran’s Caustic Soda Price: A Must-Read Comprehensive Guide to Improve Your Knowledge
Iranian Caustic Soda Pricing Analysis: Overview
Understanding Global Asphalt Prices: A Must-Read Comprehensive Guide for Better Comprehension
Cost Analysis of Caustic Soda: Overview
For more information, please click on each product listed below.
CAUSTIC SODA Products
CAUSTIC SODA FLAKES
INTRODUCTION OF CAUSTIC SODA FLAKES Sodium hydroxide, commonly known as Caustic Soda Flakes or NaOH,
CAUSTIC SODA LIQUID
INTRODUCTION OF CAUSTIC SODA LIQUID Caustic Soda Liquid is an important and versatile chemical that
Frequently Asked Questions About CAUSTIC SODA
Initially, soap manufacturers produced sodium hydroxide or Caustic Soda. The Yemeni ruler Al-Muzaffar Yusuf Ibn Umar Ibn Ali Ibn Rasul authored the book "Fi Fanon Man Al-Sunnah" in which Al-Mukhtar, a scientist from the 13th century, discovered a formula for producing sodium hydroxide. Henry Ernest Stapleton (1878-1962), an English scientist and archaeologist, has offered proof that Mohammad Zakaria Razi was familiar with sodium hydroxide. The sodium hydroxide is produced by running water through a combination of lye, which is saltwort ash, and quicklime. European soap manufacturers followed the same guidelines. Other accounts indicate that Humphry Davy developed sodium hydroxide in 1807. Unknown is the test performed to find this chemical.
Caustic Soda is an odorless and white chemical compound, and the solution of Caustic Soda in alkaline water is extremely corrosive, making it useful in a variety of industries. Sodium hydroxide, sometimes known as Caustic Soda, is one of the most commonly used chemicals in industry. Due to its alkalinity and fat-dissolving characteristics, Caustic Soda is used in several industries. Consequently, this chemical compound is often called an industrial degreaser or degreaser. 111 grams of Caustic Soda are soluble in 100 milliliters of water at 20 degrees Celsius. Increasing the temperature naturally enhances the solubility of solid Caustic Soda in water. Equally soluble in organic solvents such as ethanol, methanol, and glycerin is sodium hydroxide.
Given that Caustic Soda is a material with a strong corrosive capacity or an alkaline chemical composition, a number of industrial applications have been identified for it, including:
- The production of organic chemicals.
- Manufacturing cosmetics such as soaps and exfoliating masks.
- Production of all types of industrial detergents based on the solubility of lipids by sodium hydroxide.
- Raw material used in the manufacture of all pigments and colors in the dye industry.
- Bleaching of textile-related fabrics.
- Manufacturing of various types of ceramics and glasses.
- Defined food industry procedures.
- Fuel cell production.
- PH adjustment factor in water filtration and water hardness reduction.
- Eliminating acid from chemicals used in the petrochemical sector and oil and gas refineries
Certainly, it is not inappropriate to say that its primary uses are in the manufacture of aluminum for the steel and metallurgy sectors, the manufacturing of paper pulp and the bleaching of paper and chemical industries.
1- High alkalinity is one of the features of Caustic Soda. As a result, it is used as a pH regulator.
2- The melting point and density of Caustic Soda Flakes are 318 degrees Celsius and 2.13 grams per cubic centimeter, respectively.
3- In solid form, sodium hydroxide is white and odorless, and it dissolves in hot water. Therefore, entering the water causes its temperature to rise.
4- One of the characteristics of Caustic Soda is its exceptional capacity to absorb water and carbon dioxide. This necessitates packaging that is resistant to moisture.
5- Sodium hydroxide, sometimes known as Caustic Soda, is a strong alkali. Even if there is no danger of explosion or fire. The reaction between sodium hydroxide and acids, such as hydrochloric acid, neutralizes and generates a great deal of heat.
6- It causes metals such as aluminum, tin, and zinc to corrode. It creates hydrogen, which has the ability to act as an explosive gas, throughout this process.
7- They are very absorbent for water and carbon dioxide or sulfur dioxide in the air.
8- When Caustic Soda Liquid is diluted, a substantial quantity of heat is produced. Because this rapidly creates great heat, and if water is put into it carelessly, the resultant solution may splash, care should be used while adding water.
A lye is a metal hydroxide usually made by leaching wood ashes, or a highly soluble alkali that produces caustic basic solutions. Most often, the term "lye" refers to sodium hydroxide or Caustic Soda NaOH. Currently, lye is produced commercially utilizing a membrane cell chlor-alkali method. Soap has always been made using lye as a main component.
Numerous enterprises make substantial use of Caustic Soda. Consequently, its manufacturing rate is increasing everyday throughout the majority of the world. Oceania, China, the United States, Germany, etc. are now among the top producers of this commodity. China is the only country in Asia with a considerable sodium hydroxide market, and it is one of the leading producers of this chemical. The energy-intensive and expensive nature of Caustic Soda production has a significant impact on market growth and price volatility. Nonetheless, as production in other industries grows, so will demand for this material.
China will account for over sixty percent of the sodium hydroxide market by 2031, according to PetroNaft Company's research team. This country has committed around $10 million to develop a Caustic Soda production factory. Also, the demand for sodium hydroxide or Caustic Soda is gradually increasing on the U.S. market, and it is projected that sales of this material will reach their peak around 2030. surpass 81 million dollars in worth. It should be underlined that Germany's chemical manufacturing business is one of the world's most well-known and has always developed substantially. According to the report, in 2021 the German market for Caustic Soda generated around $1.3 billion.
To buy the Caustic Soda, you can click "ORDER NOW" below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
In the drilling industry, the following situations call for sodium hydroxide with the formula NaOH, often known as CAUSTIC SODA:
Calcium and magnesium cations may precipitate as Ca(oh)2 and Mg(oh)2 when the pH of drilling muds is increased in order to prevent corrosion and minimize pollution caused by calcium or magnesium in muds.
To produce the appropriate alkaline environment in order to distribute the particles and boost the production of bentonite muds.
Due to its total solubility in water, it will not leave any deposits on the pipes and equipment used for mud-related tasks.
It serves a crucial function in drilling by enabling and conserving drilling mud.
The leather industry is one of the rare sectors in which a natural raw material undergoes many phases of transformation into a standard product. In the past, it took around three months to complete all phases of leather manufacture using the old method. During the degreasing process, Caustic Soda is employed in the leather industry following depilation and removal of wool and non-fibrous proteins from the surface of the skin by lime. Some skins, like sheepskin, contain fat, which must be removed prior to tanning. As is well known, Caustic Soda has very corrosive and fat-dissolving qualities; hence, it is used to remove fat in leather and tannery factories and workshops.
To produce one ton of paper, 5 cubic meters of wood are needed, which is obtained from 50 trees. Recovering 1 ton of waste paper means saving 17 trees. Recovery of waste paper preserves the environment and forests and prevents energy waste.
Sodium hydroxide, or Caustic Soda, is a widely used chemical substance in various industries. Sodium hydroxide, or Caustic Soda, is used as the main chemical substance in ink removal.
Types of ink and their properties:
-Oily ink:
It is hydrophobic.
It is difficult to separate from the paper.
It is difficult to wash off.
Washing water pollutes the environment.
Its average particle size is 5 to 10 μm.
It is suitable for the flotation process.
It has a low density.
-Aqueous ink:
It simply peels off the paper.
It is easily removed by washing.
Washing water does not pollute the environment.
Its average particle size is smaller than 1 μm.
It is used for the washing process.
The nature of ink particles:
Typical ink particles consist of the following materials:
Pigment
Adhesive agents (oily or aqueous).
Additives (organic acid, resins).
Covering and clarifying
Ink removal for paper recycling is done in the following ways:
1-Ink removal by washing
2-Ink removal by flotation
- Temperature:
Less than 40 degrees Celsius is the ideal storage temperature for Caustic Soda. It is vital to note that the liquid viscosity of this product (Caustic Soda Liquid 50%) significantly rises at low temperatures. Consequently, there may be issues with the product's transportation and pumping. In cold weather, it is essential to heat product transmission lines to prevent obstructions in the transmission pipes. This may be accomplished with 103 kPa of steam flow or with electric heating. As a consequence, Caustic Soda storage warehouses must always be equipped with temperature control so that the temperature stays consistent between 29 and 38 degrees Celsius. These controllers (thermocouples) provide a warning signal if the temperature increases.
- Tank and bag assortment:
When storing Caustic Soda, three significant considerations must be taken into account: 1- The hazardous and corrosive character of this product, which may be very hazardous to personnel. 2- The viscosity of the liquid form of this substance rises fast at temperatures below 16 degrees Celsius, hence the storage temperature must be above this threshold. 3- The weight of 50 percent sodium hydroxide liquid is 1.5 times that of water (with the same volume).
Solid Caustic Soda is often kept in thin steel drums; using aluminum, tin, zinc and its alloys, chromium, or lead may cause severe damage. In aluminum tanks, for instance, the interaction of hydrogen and oxygen in the presence of heat results in the production of extremely flammable hydrogen gas.
Caustic Soda Flakes are often kept in paper or PVC-coated paper bags. In the next sections, we will examine the maintenance requirements for each of the following situations:
1- Carbonic acid in a bag:
To preserve the stability of the product, the bags must constantly be stored in a warehouse with a dehumidifier and air conditioner to eliminate moisture and chill the air.
If outdoor storage is inevitable, the bags should be coated with black plastic to prevent exposure to moisture and sunlight.
Prevent absorption of moisture and carbon dioxide by preventing direct contact with air.
2- Caustic Soda in the barrel:
Drums should be stored upright and ideally inside to avoid corrosion caused by surface water. During the maintenance time, ensure that the valves stay closed. Cover the drums with a waterproof coating if you are storing them outdoors. Maintain a constantly dry warehouse.
Because Caustic Soda is very corrosive and highly irritating, workers must wear protective coverings at all stages of working with it.
Due to its corrosive qualities, contact with Caustic Soda causes severe irritation and damage to the skin and eyes. In the event of bodily contact with sodium hydroxide powder, first remove the powder with a dry towel and then wash the affected area with water for 30 minutes. Note that when Caustic Soda is combined with water, it turns corrosive; thus, ensure that all of the powder is removed from the skin's surface before washing with water. People may sometimes use wet wipes to remove the powder from the skin's surface from the very beginning; nevertheless, the wipes' moisture might react with Caustic Soda and cause severe injury to the body.
In the event of contact with Caustic Soda Liquid, you must first remove the solution from the skin's surface with a dry towel and then wash the skin with water for 30 minutes, until the solution has been entirely removed from the skin's surface.
Depending on the concentration of the infected fluid, sodium hydroxide might cause blindness if it enters the eye. If your eyes get tainted with Caustic Soda, you should immediately contact a professional and wash your eyes with water until the surface of your eyes is entirely clean.
Depending on the vapor concentration, inhaling sodium hydroxide may result in minor to severe harm, including sneezing, sore throat, runny nose, and lung irritation.
- The harmful effects of Caustic Soda on the skin:
Due to its strong reactivity, Caustic Soda may eliminate fat from a variety of surfaces. Unfortunately, one of the worst consequences of Caustic Soda Flakes on the skin is producing severe burns, which are common in households.
- Caustic Soda is hazardous to the eyes:
As soon as Caustic Soda joins water, exothermic reactions occur and hazardous gases are released into the atmosphere. After entering the eyes, this gas causes significant visual impairment as well as eye burns and redness.
- Caustic Soda Flakes' threats to the respiratory system:
The Caustic Soda Flakes and water solution should never be used in enclosed or poorly ventilated areas. After interacting with water and other compounds on the target surface, these substances form poisonous gas, which, when inhaled, causes harm to the lungs and respiratory tract.
- Dangers of Caustic Soda Flakes to the digestive tract:
One of the worst possible outcomes of ingesting Caustic Soda Flakes is for these chemicals to enter the digestive tract. These very corrosive chemicals attack the lipids in the digestive system and cause serious burns.
In the event of contact with the body, just water is required to remove Caustic Soda; washing and neutralizing chemicals should not be used. Immediately after beginning to wash with water, medical facilities should be called to acquire the essential advice. Continue washing for 30 minutes unless instructed otherwise by a medical professional.
When a person does not wash after coming into touch with water, irreversible harm is caused.
This compound is not classified as a carcinogen by the Department of Health and Human Services (DHHS), International Agency for Research on Cancer (IARC), or Environmental Protection Agency (EPA). Corrosiveness and tissue damage have also been linked to esophageal cancer; however, this chemical is not a carcinogen.
Ask the maker or supplier of the Caustic Soda Flakes you bought for the MSDS.
- Eye contact:
Immediately flush the eyes with a low-pressure eyewash fountain. After washing the hands thoroughly, keep the eyelids open and rinse the eyes with water for 30 minutes; discontinue rinsing with water only if instructed otherwise by a professional.
- Dermal contact:
Continuously wash the affected skin with water for thirty minutes. Remove your clothing before bathing. If your eyes are not polluted, do not remove your safety glasses until your head and hands have been thoroughly cleansed. Because Caustic Soda may dissolve in water and enter the eye after cleaning. Burns caused by this substance may not manifest until 24 to 48 years after the incident.
- Consuming:
In the event of swallowing, you should immediately consume a large amount of water or milk (about 240 ml) and contact a medical facility. If the poisoned individual has lost consciousness, he should not be forced to consume anything. Avoid vomiting induction.
- Breath in:
In the event of inhalation of Caustic Soda Flakes vapor, immediately transport the victim to fresh air and contact medical professionals. If the poisoned individual is not breathing, give him artificial respiration; if he has difficulty breathing, give him an oxygen capsule (should be done by experienced people).
-Dress:
Before reuse, wash soiled garments and coverings completely. Leather products such as shoes, belts, and watchbands should be cleaned.
In the event of contamination with Caustic Soda, contacting medical emergency services as soon as possible is crucial.
Caustic Soda Flakes react vigorously with numerous organic and inorganic substances, such as strong acids, nitroaromatics, nitroparaffin compounds, organ halogens, glycols, and organic peroxides; on some plastics, such as PET (Soda bottle), they leave an impression and pierce them; when combined with certain metals, they generate hydrogen gas.
The worldwide standard for packaging Caustic Soda Flakes in 25-kilogram multi-layer bags is to prevent the flakes from reacting with carbon dioxide gas and absorbing moisture. The 25 kg bags are placed on pallets or loaded into jumbo bags so that they may be moved more easily by forklifts and cranes, particularly for export.
- Significant chemical changes occur.
- Explosions may be caused by strong shock or heat.
- It is often stable but becomes unstable when heated.
- This substance primarily affects human and animal tissues, including leather, skin, and eyes.
- It interacts efficiently with reducing sugars such as maltose, fructose, and galactose, producing carbon monoxide.
First, weigh and record the weight of 1 cc of the Caustic Soda Liquid sample. Then, dilute it to the desired level (100 cc) with water and transfer 20 cc of the diluted solution to an Erlenmeyer flask. Then, add two drops of phenolphthalein Now, pour a certain quantity of 0.1 normal hydrochloric acid into a burette. Then, gently open the burette and pour the hydrochloric acid drop by drop into the Erlenmeyer flask, while continually shaking the solution in the Erlenmeyer flask. Perform the mixing procedure until the indicator's color changes from purple to colorless and remains unchanged. Now, based on the quantity of acid eaten, the sample's concentration may be determined.
Liquid sodium hydroxide or Caustic Soda Liquid is a potentially dangerous chemical. It may be harmful if it comes into contact with the skin, is ingested, or is inhaled. Consuming liquid sodium hydroxide may result in severe burns, vomiting, nausea, diarrhea, chest and stomach discomfort, and swallowing difficulties. Additionally, damage to the lips, throat, and stomach occurs rapidly. It may irritate the upper respiratory tract severely, resulting in coughing, burning, and trouble breathing.
The adverse effects of Caustic Soda depend on a number of circumstances, including the quantity of sodium hydroxide, the duration of exposure, and whether the substance was touched, ingested, or breathed.
Extremely high concentrations of liquid sodium hydroxide may result in permanent harm or death when in contact with the eyes, skin, digestive tract, or lungs. Dermatitis may result from prolonged or recurrent skin contact. Inhaling Caustic Soda fumes repeatedly might cause lifelong lung injury.
To utilize Caustic Soda Liquid, anti-acid gloves, acid-resistant clothing, and acid-resistant footwear must be used.
- Eye contact: Use special chemical goggles.
- Occupational contact: Gloves, work clothing, and work shoes resistant to chemicals, as well as a shower and eye protection, are required in locations where this material is present.
- Respiratory contact: When dealing with chemicals, use appropriate masks.
To know the price range of Caustic Soda, you can click "ORDER NOW" below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
To download the MSDS of Caustic Soda, you can click the "INFORMATION REQUEST AND PRODUCT SAMPLE" button below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
To download the TDS of Caustic Soda, you can click the "INFORMATION REQUEST AND PRODUCT SAMPLE" button below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
To download the specification PDF of Caustic Soda, you can click the "INFORMATION REQUEST AND PRODUCT SAMPLE" button below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
To know the price of caustic soda in Iran, you can click "ORDER NOW" below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
To know the price of caustic soda in China, you can click "ORDER NOW" below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
To know the price of caustic soda in India, you can click "ORDER NOW" below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.
To know the price of caustic soda in the UAE, you can click "ORDER NOW" below the product photos. We will send you the necessary information as soon as possible, or use the "ANY QUESTION?" form on the bottom right of the page.







Dear all, I’m interested to understand, how does caustic soda participate in the saponification process?
Hello and greetings! That’s a great question which touches on the chemical processes involved in soap making.
In saponification, caustic soda, also known as sodium hydroxide, plays a crucial role. When combined with a triglyceride (fat or oil), a reaction occurs. This reaction breaks down the triglyceride into its constituent parts – glycerol and fatty acid salts, the latter of which are what we commonly refer to as “soap”.
In more technical terms, the hydroxide ions (OH-) from the caustic soda break the ester bonds in the triglycerides. This causes the molecule to separate into glycerol and fatty acids. The sodium ions (Na+) from the caustic soda then bond with the fatty acids, creating soap.
It’s a fascinating process and a classic example of how chemistry is a part of everyday life. I hope this helps provide a better understanding!
Dear sir or madam,
We are from the company ProClean Enterprises, and we have been purchasing your product Caustic Soda, Grade Flake. We are thoroughly satisfied with both the quality of the product and your services. Despite our satisfaction, we have one query we’d like to address for further clarification. Can you elaborate on the precautions needed to store Caustic Soda properly, specifically in higher temperature environments?
Hello,
Thank you for your good question and your kind words about our product and service. Caustic Soda is a strong alkali, and it’s important to handle and store it correctly to ensure safety and maintain product quality. Even though it is not directly sensitive to temperature, higher temperatures can accelerate its absorption of moisture from the air, causing it to become more liquid and potent. It should be kept in a cool, dry place, ideally below 30°C. Ensure that the storage container is airtight and resistant to caustic soda, like HDPE (High-Density Polyethylene). Always use protective gear when handling it and keep it out of reach of unauthorized individuals. Your health and safety are of the utmost importance to us.
Hello, we are from Company PowerPipe, and we have purchased Grade A caustic soda from you. We are fully satisfied with both the quality and your services. We were wondering how best to store this product to ensure it maintains its efficacy over time?”
Hello dear, thank you for your kind words and your question. Storage of caustic soda is crucial for maintaining its efficacy. It should be stored in a cool, dry place away from any source of moisture, as it reacts readily with water. Additionally, it should be kept in a tightly sealed container to avoid contact with air. The container material should be resistant to alkali such as HDPE (High-density polyethylene) or PP (Polypropylene). It’s also recommended to keep it out of the reach of children and pets due to its corrosive nature. Safety should always be your priority when dealing with caustic soda.
please explain about naoh molar mass
Sure, I’d be happy to help you out!
NaOH is the chemical formula for sodium hydroxide, which is a common ingredient in many petroleum products. The molar mass of NaOH can be calculated by adding the atomic masses of sodium (Na), oxygen (O), and hydrogen (H) that make up the compound.
The atomic masses of these elements are:
Sodium (Na) = 22.99 g/mol
Oxygen (O) = 15.99 g/mol
Hydrogen (H) = 1.01 g/mol
So, to calculate the molar mass of NaOH:
Molar mass = (Na atomic mass) + (O atomic mass) + (H atomic mass)
Molar mass = (22.99 g/mol) + (15.99 g/mol) + (1.01 g/mol)
Molar mass = 39.99 g/mol
Therefore, the molar mass of NaOH is 39.99 grams per mole (g/mol). I hope this helps! If you have any other questions or concerns, please don’t hesitate to ask.
good day
I wanted to ask about the price of caustic soda
Thank you for your question
To know the exact price, please contact us via the email below
[email protected]
I am not sure where you are getting your information, but good topic.
I needs to spend some time learning more or understanding more. Thanks for wonderful info I was looking for this info for my mission.
Thank you for your comment and interest in our website. We strive to provide accurate and up-to-date information about our petroleum products to our customers. If you have any specific questions or concerns about our products, please do not hesitate to contact us. We would be more than happy to assist you and provide you with any additional information you may need. Additionally, we regularly update our website with new information and resources, so please check back often for the latest updates. Thank you again for your feedback, and we look forward to serving you.
I think the admin of this site is in fact working hard in favor of his web page, because here every information is quality based information.
Thank you for your kind words! We are dedicated to providing our customers with high-quality information about our products and services. We believe that informed customers make better purchasing decisions, which is why we strive to ensure that our website is filled with informative and helpful content.
If you have any questions or concerns about our products or services, please don’t hesitate to contact us. We are always happy to help and are committed to providing the best customer service possible.
Also, if you found our website helpful, please consider leaving us a review or sharing it with your friends and family. Your support is greatly appreciated and helps us to continue providing high-quality products and services.
Thank you again for your kind words, and we look forward to serving you in the future!
I’ve been exploring for a bit for any high-quality articles or weblog posts in this kind of house . Exploring in Yahoo I at last stumbled upon this website. Reading this information So i am happy to exhibit that I’ve a very good uncanny feeling I discovered just what I needed.
I so much surely will make sure to do not overlook this website and give it a look on a continuing basis.
Thank you for taking the time to visit our website and leaving your feedback. We are delighted to hear that you found our content helpful and valuable. As a leading provider of petroleum products, we take pride in offering high-quality articles and weblog posts to our visitors.
We also appreciate your commitment to staying up-to-date with our website and checking back regularly. Our team is dedicated to providing the best possible user experience, and we are thrilled to have earned your loyalty.
If you have any questions or concerns, please do not hesitate to reach out to us. We are always happy to help and look forward to serving you in the future. Thank you again for your kind words and support!
Your web site provided us with useful information to work on. You have done an impressive task and our whole community will likely be grateful to you.
Thank you for taking the time to leave a comment and for your kind words. We strive to provide our customers with the most up-to-date and useful information about our petroleum products. It is great to hear that our efforts have been useful for you and your community. We appreciate your support and hope to continue serving you in the future. Don’t hesitate to contact us if you have any questions or need further assistance.
Thanks in favor of sharing such a pleasant idea, article is pleasant, thats why i have read it fully
You’re welcome! I’m glad that you found the idea and article pleasant and that you read it fully. If you have any questions or want to discuss any related topics further, feel free to let me know!
Hello, how does caustic soda get made?
Good day and welcomes! Sodium chloride is commonly electrolyzed to produce caustic soda.
Hello, can caustic soda be used to create biodiesel?
Good day and welcomes! Indeed, caustic soda may be utilized as a catalyst in the biodiesel synthesis process.
Thank you for the useful information; what are the many types of caustic soda?
You are very welcome! Caustic soda may be found as flakes, pellets, or as a solution.
Dear Everyone, Is caustic soda a dangerous substance?
Thank you for your feedback. Because of its corrosive nature, caustic soda is classified as a hazardous substance.
Hey there, Is caustic soda equivalent to sodium hydroxide?
Thank you for your feedback. Caustic soda is a synonym for sodium hydroxide.
Good day, what precautions should I take while dealing caustic soda?
Thank you for your inquiry. While handling caustic soda, it is essential to use protective clothes such as gloves and goggles since it may inflict serious burns and eye damage.
Greetings, Madam. What are the effects of utilizing caustic soda on the environment?
Thank you for your feedback. Caustic soda manufacturing and usage may have negative environmental consequences such as air pollution and the formation of hazardous waste. Nevertheless, appropriate handling and disposal may lessen the effects.
Hello, what are some common home applications for caustic soda?
Thank you for your inquiry. Caustic soda may be used to clean ovens and stovetops as well as unclog drains.
What are the risks of being exposed to caustic soda, please?
Thank you for your inquiry. Caustic soda exposure may result in chemical burns, skin irritation, and eye damage.
Hello, could you please explain me what caustic soda is used for?
Good day and welcomes! Caustic soda is utilized in a wide range of applications, including soap production, water purification, and cleaning agents.